
The Peacekeepers Within
Introduction: defense and restraint in the immune system
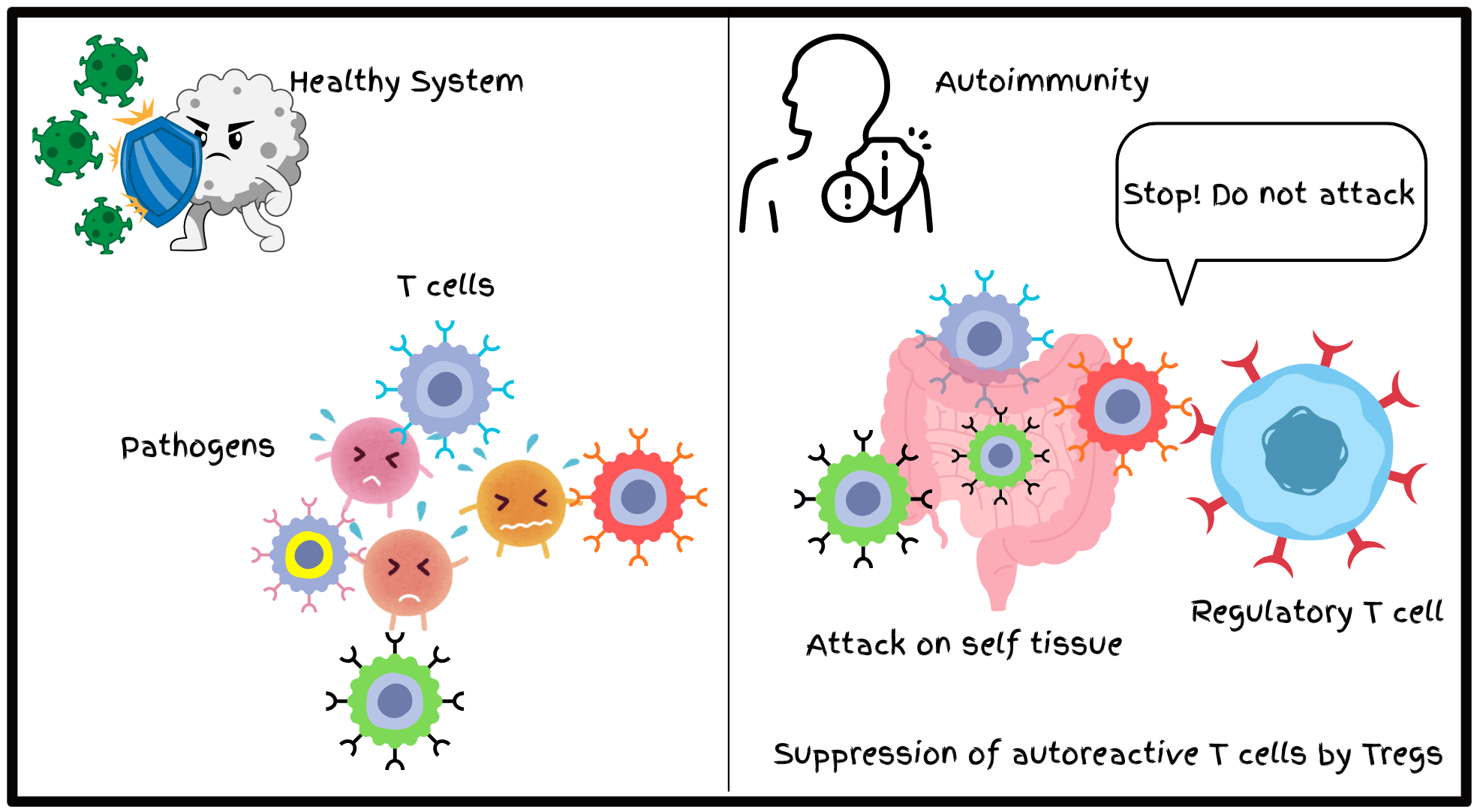
Multicellular organisms exist in constant exposure to a wide array of microbial threats. To counter these threats, vertebrates have evolved an immune system which is capable of pathogen detection and elimination by employing both innate and adaptive lines of defense. Innate immunity is the first line of defense to fight any pathogenic invasion immediately while adaptive immunity is the second line of defense that is slower and is characterized by ‘memory’ for faster future encounters. At the core of adaptive immunity are T lymphocytes that recognize antigens through T cell receptors (TCRs). These receptors exhibit an extraordinary diversity as they are generated via a gene rearrangement mechanism commonly known as somatic recombination. TCRs thus have the capability to recognize diverse set of antigenic peptides processed and presented by antigen presenting cells (APCs). However, the same random TCR rearrangement that allows T cells to recognize a wide range of antigens also creates an inherent risk: some TCRs can recognize self-antigens, potentially leading to autoimmunity - a condition where T cells start to attack our own tissues. However, under normal conditions, these autoreactive T cells are kept under check, and they are not allowed to cause damage to the self tissues - a process commonly known as immune tolerance. This raises an important question: what molecular mechanisms keep self-reactive T cells in check in the periphery? Are specific cell types responsible? Are soluble factors involved? Or is it achieved through a combination of cellular and molecular forces?
It took decades to find out the answer to this mystery. Through a series of experiments and combined efforts of three scientists- Shimon Sakaguchi, Mary E. Brunkow, and Fred Ramsdell- another regulatory layer to T cell function known as peripheral immune tolerance was uncovered. The 2025 Nobel Prize in Physiology or Medicine was awarded to Shimon Sakaguchi, Mary E. Brunkow, and Fred Ramsdell, who unwrapped the cellular and genetic architecture of ‘peripheral immune tolerance’. Their discoveries reshaped immunology, providing a conceptual framework for understanding autoimmunity, allergy, transplantation, and cancer. The following sections trace the scientific journey that led to these transformative insights.
T cell development and tolerance mechanisms
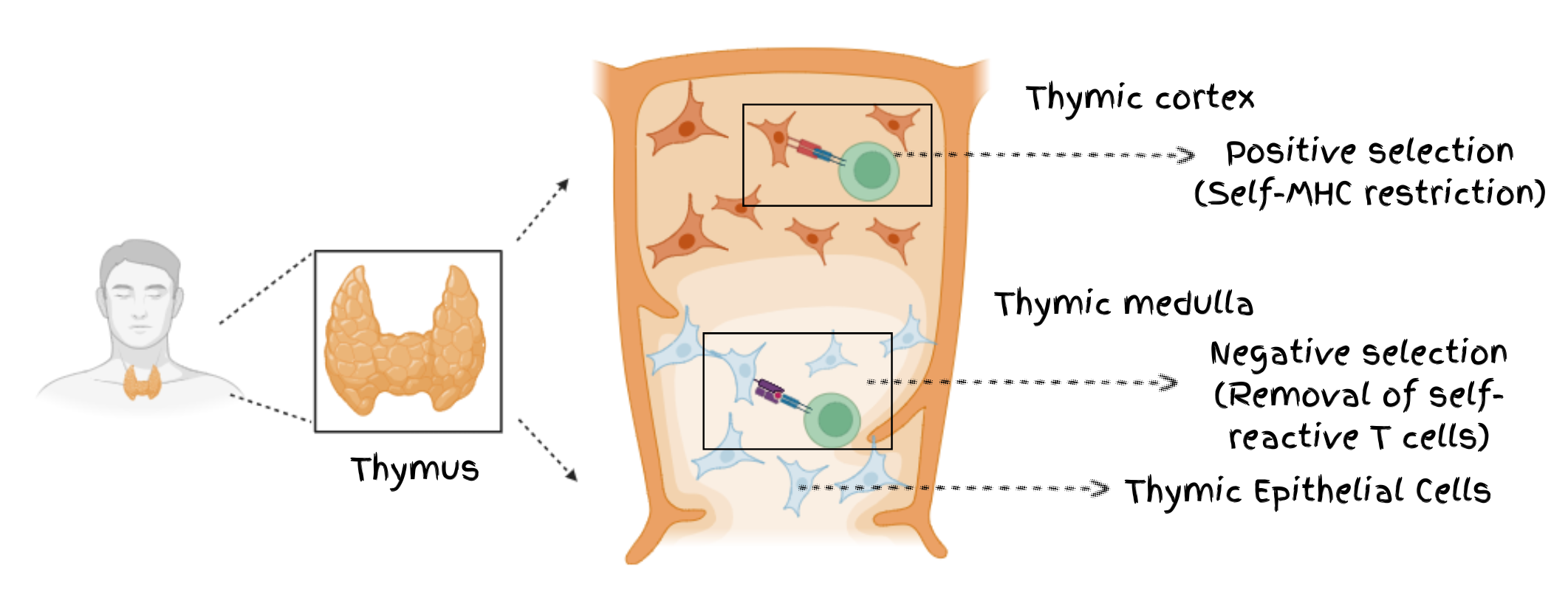
T cells originate from common lymphoid progenitors (CLPs) in the bone marrow, but they complete their maturation in the thymus. Thymus is a small, bilobed organ located above the heart and contains two major compartments- the cortex and the medulla- whose organization is essential for proper T cell maturation and TCR diversification. Once T cell progenitors enter the thymic environment, they progress through a series of orderly developmental stages that are defined largely by the expression patterns of surface proteins CD4 and CD8.
Early in their development, T cell precursors first lose the potential to differentiate into other lineages, such as B cells. They then enter the double-negative (DN) stage, characterized by the absence of both CD4 and CD8. As they mature, they transition into the double-positive (DP) stage, expressing both CD4 and CD8 simultaneously. Ultimately, they differentiate into single-positive (SP) thymocytes, expressing either CD4 or CD8, which determines their functional fate in the periphery .
During their developmental transitions, T cells have to cross 2 major checkpoints. First one is positive selection which ensures that only those T cells which are self-major histocompatibility complex (MHC) restricted survive; while second one is negative selection which facilitates the elimination of T cells that react too strongly to the molecules present on the body “ s own cells, commonly known as self-antigens . This process helps in preventing autoreactive clones from entering the periphery and is also known as ‘central immune tolerance’.
Once T cells maturation is complete, they exit the thymus and migrate to secondary lymphoid organs such as the spleen and lymph nodes. These are the sites where they encounter APCs, recognize antigenic peptides, and become activated.
Despite the stringent selection events in the thymus, some autoreactive T cells may escape central tolerance and enter the periphery. What happens then? How does the body prevent these cells from attacking self-tissues?
This is where peripheral tolerance comes into play. Peripheral tolerance ensures that autoreactive T cells that reach the peripheral tissues are either eliminated or functionally restrained so that they do not cause self-damage. Peripheral tolerance is accomplished through several mechanisms : promoting the activity of regulatory T cells, inducing anergy (a state in which T cells become unresponsive), or triggering clonal deletion, which removes these harmful T cells altogether.
But where do these regulatory T cells come from? Do they also develop in the thymus? Yes, a fraction of regulatory T cells (commonly known as Tregs) also develop in the thymus. During thymic selection, when the TCR on developing CD4 T cells engages with the cognate receptor (MHC-peptide complex) present on an APC, it triggers downstream TCR signalling. Surprisingly, the strength of this TCR signalling determines the fate of the CD4⁺ T cell to either become a conventional CD4⁺ T cell or a regulatory T cell (Treg). Low-affinity TCR signals favor development into conventional CD4⁺ T cells, while intermediate-strength signals promote commitment to the Treg lineage. However, if a thymocyte receives very strong TCR signals, it undergoes negative selection and is deleted from the developing repertoire.
Controversial suppressor T cell hypothesis
For a long time, the thymus was seen as the sole guardian against autoimmunity. The occurrence of autoimmune diseases was largely interpreted as failures of thymic selection. However, the gradual accumulation of experimental evidence across the globe challenged this view. For example, people observed that in some animals despite having intact thymic architecture, they are prone to autoimmune symptoms.
Not only this, reports from the 1970s hinted at a different layer of regulation which was the “suppressor T cells” . These studies suggested that certain T cells could actively restrain immune responses, but reproducibility was poor, and no reliable molecular markers existed to prove their presence. As a result, the concept remained scientifically marginalized, and the observations were considered as misinterpreted data.
Amid this skepticism, Shimon Sakaguchi showed his determination and pursued the unconventional idea that a subset of peripheral T cells might actively suppress autoreactive clones. Building on the hints from earlier studies, he hypothesized that these cells could provide a critical layer of peripheral tolerance, setting the stage for experiments that would ultimately redefine our understanding of immune regulation.
The neonatal thymectomy paradox
According to the prevailing thoughts at that time, scientists predicted that thymectomy would lead to immunodeficiency, as the thymus was thought to be the sole source of functional T cells. Yet the results defied expectation. To their surprise, they observed that removal of thymus (thymectomy) in newborn mice at day 3 after birth did not lead to a weakened immune system. Instead, the mice developed severe multi-organ autoimmune disease, including thyroiditis , gastritis , and oophoritis . This was very surprising! It suggested that thymus was not only responsible for the production of effector T cells but also a regulatory T cell population that restrains self-reactive T cells in the periphery.
So, to further test this hypothesis, Sakaguchi transferred a specific fraction of peripheral T cells from genetically identical, healthy mice into thymectomized mice recipients . Remarkably, the autoimmune symptoms were reversed, providing the first functional evidence that peripheral T cells actively enforce self-tolerance.
This discovery shifted the conventional paradigm. The immune tolerance was not solely established through negative selection in the thymus but required active immune suppression in the periphery. It laid the foundation for identifying the T cell subset responsible for this critical regulatory role - the regulatory T cells (Tregs).
Sakaguchi: Discovery of CD25⁺ regulatory T cells
Motivated by the observations from thymectomy experiments, Sakaguchi desired to identify the cellular population responsible for maintaining peripheral tolerance. He focused on a rare subset of CD4⁺ T cells expressing CD25 (a receptor present on CD4⁺ T cells that binds to IL-2 - a critical growth factor for T cells), which had previously been overlooked and largely dismissed as merely activated effector cells. But how do we figure out the exact role of these CD25 expressing T cells? Sakaguchi took a clever approach. To test the function of this T cell subset, he selectively depleted CD25⁺ T cells from healthy mice and transferred these cells in athymic ‘nude’ mice that are immunodeficient. The results were striking. These mice developed multi-organ autoimmunity, including tissues such as thyroid, pancreas, and stomach being attacked . Interestingly, transfer of CD4 CD25 T cells within a limited period after the transfer of CD4 CD25 T cells prevented autoimmune responses. This provided definitive evidence that CD25⁺ T cells were not ordinary effectors but instead a regulatory population critical for restraining self-reactive T cell clones. However, the story does not end here. The problem of not having a proper genetic marker for regulatory T cells was still there and was resolved only partially, as CD25 is expressed on activated effector T cells, though at a lower level. Thus, it limited its specificity and was not a perfect marker for the identification of this newly emerging regulatory T cells .
The field thus required a precise molecular identifier to define Tregs uniquely, setting the stage for the next critical discovery in the story of immune regulation.
Brunkow and Ramsdell: Solving the scurfy mouse mystery
While Sakaguchi was defining the cellular basis of immune regulation, Mary E. Brunkow and Fred Ramsdell were investigating a parallel line of puzzle of a peculiar mouse strain known as the scurfy mouse. These mice exhibited unique features like scaly skin, enlarged lymphoid organs, and severe multi-organ inflammation, with a pattern of male-specific lethality suggesting an X-linked mutation. The animals died early due to overwhelming autoimmunity, but the precise gene responsible remained unknown.
Brunkow and Ramsdell accepted the challenge of understanding the genetics behind scurfy phenotype. As limited molecular tools were available that time, they utilized the positional cloning techniques, genetic mapping, candidate gene sequencing and combinatorial breeding strategies to address this question. After years of meticulous work, they identified an insertion of two-nucleotides in a previously uncharacterized gene. It is worth noting that Brunkow and Ramsdell had to painstakingly screen multiple genes before reaching to the gene responsible for scurfy phenotype. This marks their dedication and efforts which they had put up into. Biologically, this mutation caused a frameshift and premature stop codon and thus abolishing the gene’s function. Based on sequence homology with other forkhead box genes, they named it FOXP3 (Forkhead box P3) .
The significance of this discovery became even more apparent when Brunkow and Ramsdell observed striking similarities between phenotype of scurfy mouse and human IPEX syndrome (Immune dysregulation, Polyendocrinopathy, Enteropathy, X-linked) and they started to suspect that IPEX is the human equivalent of scurfy mouse. Children with IPEX suffered from early-onset chronic diarrhoea, autoimmune enteropathy, and severe inflammatory skin rashes, often leading to death in infancy. Genetic analysis revealed mutations in FOXP3 , paralleling the scurfy mouse defect .
This convergence of mouse genetics and human pathology established FOXP3 as essential for immune tolerance. It further set the stage for linking two previously independent lines of investigation- Sakaguchi’s cellular discoveries and Brunkow/Ramsdell’s search for a definitive genetic regulator of autoimmune disease in mice and humans.
FOXP3: The master regulator of Tregs
For years, Sakaguchi’s identification of CD25⁺ regulatory T cells and Brunkow and Ramsdell’s discovery of FOXP3 had proceeded along parallel tracks. The unifying breakthrough came when Sakaguchi demonstrated that Foxp3 is selectively expressed in CD4⁺CD25⁺ Tregs . Introducing Foxp3 into conventional CD4⁺T cells further led to the conversion of conventional CD4⁺ T cells into cells with suppressive function . Not only this, the scurfy mice having mutations in Foxp3 were found to be completely devoid of functional Tregs, whereas mice that overexpressed Foxp3 carried excessive Treg cells .
This finding established FOXP3 as the master transcription factor defining the Treg lineage. What had once been a controversial concept of suppressor T cells was now a well-defined biological entity. The concept of Tregs explained many long-standing questions including how the immune system maintains tolerance even after thymic selection.
Mechanisms of Treg-mediated immune suppression
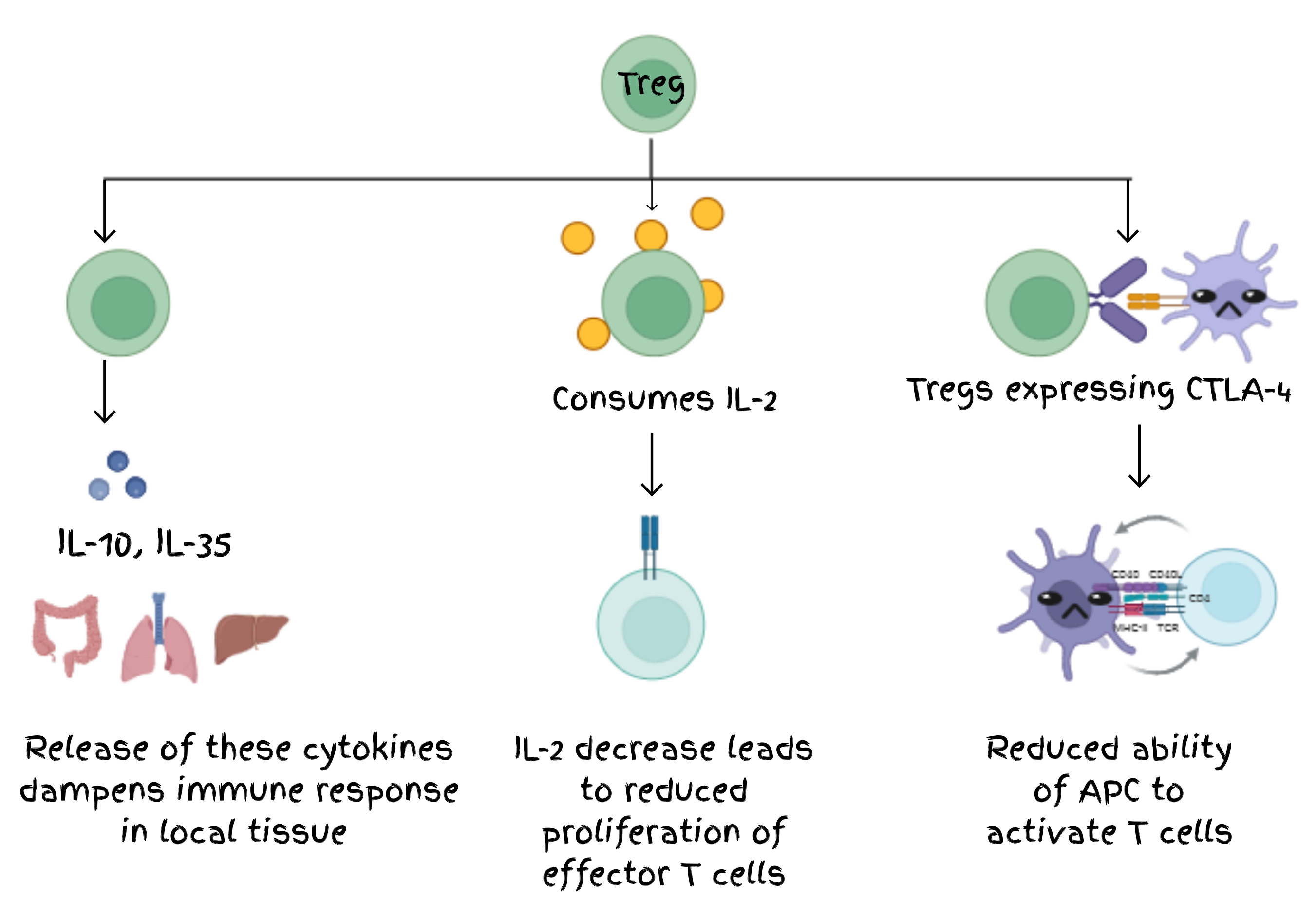
The identification of cellular and molecular basis of peripheral immune tolerance was a key discovery, but then at functional level, how exactly Tregs provide protection against self-reactivity? The discovery of FOXP3 as the master regulator sparked a wave of curiosity about the biology of Tregs. With this molecular marker, researchers could isolate Tregs and investigate how these cells actively maintain immune tolerance. Tregs were found to employ multiple, complementary mechanisms to suppress immune activation.
Tregs secrete anti-inflammatory cytokines such as IL-10, TGF-β, and IL-35, which dampen immune responses in local tissues. They consume IL-2, a critical T cell growth factor, thereby limiting the proliferation of autoreactive effector T cells. Through CTLA-4 expression, Tregs modulate antigen-presenting cells, reducing their ability to activate effector T cells. Together, these mechanisms demonstrate that immune tolerance is actively maintained and is context-dependent, rather than a passive absence of immune response .
Clinical implications of regulatory T cells
The discovery of Tregs and FOXP3 reshaped not only fundamental immunology but also the understanding of human diseases. Dysregulation of Tregs underlies a wide spectrum of pathological conditions, highlighting their central role in immune homeostasis.
Autoimmune diseases
Defective Tregs are now recognized as key contributors to disorders such as multiple sclerosis , rheumatoid arthritis , psoriasis , type 1 diabetes and others. In these conditions, the failure of immune regulation allows autoreactive effector T cells to attack self-tissues, leading to chronic inflammation and progressive organ damage. The mechanistic insights into Treg biology have opened avenues for therapeutic intervention, including low-dose IL-2 therapy to expand Treg population and engineered Treg infusions designed to restore tolerance .
Cancer
On the other hand, regulatory T cells play an opposite role in tumors by suppressing the anti-tumor immune responses. Cancer uses Tregs to create an immune suppressive shield and this helps tumor to grow and evade faster. Scientists are now trying to specifically target tumor-associated Tregs in order to alleviate their suppression of anti-tumor immune responses and to promote clearance of tumor .
Transplantation
One of the major challenges in organ transplantation is graft rejection due to alloreactive immune responses. Tregs can suppress these reactions and thus can promote long-term graft tolerance .
Taken together, by defining the molecular and cellular mechanisms governing Treg function, Sakaguchi, Brunkow, and Ramsdell provided a foundation for interventions across diverse medical fields.

Conclusion
The story of the 2025 Nobel Prize is not only about cells and genes, but it is also a testimony of scientific courage, continuous efforts, and curiosity. Sakaguchi pursued a hypothesis that many had long dismissed. Meanwhile, Brunkow and Ramsdell devoted years to unravelling the genetic mystery of the scurfy mouse. Positional cloning at the time was a formidable endeavor, yet they pursued it despite the odds and technical limitations.
Their work ultimately bridged mouse and human immunology and illuminated the central role of FOXP3 in peripheral immune tolerance. Their discovery has opened the door to innovative therapies ranging from Treg-based interventions in autoimmune diseases, allergy, and transplantation, to strategies that modulate Treg function in cancer immunotherapy. The immune system’s peacekeepers “Tregs” stand as a testament to nature’s intricate design and the enduring power of scientific inquiry.
References
- Charles A Janeway, J., Travers, P., Walport, M. & Shlomchik, M. J. Antigen recognition by T cells. in Immunobiology: The Immune System in Health and Disease. 5th edition (Garland Science, 2001).
- Charles A Janeway, J., Travers, P., Walport, M. & Shlomchik, M. J. Generation of lymphocytes in bone marrow and thymus. in Immunobiology: The Immune System in Health and Disease. 5th edition (Garland Science, 2001).
- Ashby, K. M. & Hogquist, K. A. A guide to thymic selection of T cells. Nat. Rev. Immunol. 24, 103–117 (2024).
- Gershon, R. K. & Kondo, K. Cell interactions in the induction of tolerance: the role of thymic lymphocytes. Immunology 18, 723–737 (1970).
- Kojima, A., Tanaka-Kojima, Y., Sakakura, T. & Nishizuka, Y. Spontaneous development of autoimmune thyroiditis in neonatally thymectomized mice. Lab. Investig. J. Tech. Methods Pathol. 34, 550–557 (1976).
- Penhale, W. J., Farmer, A., McKenna, R. P. & Irvine, W. J. Spontaneous thyroiditis in thymectomized and irradiated Wistar rats. Clin. Exp. Immunol. 15, 225–236 (1973).
- Tung, K. S., Smith, S., Teuscher, C., Cook, C. & Anderson, R. E. Murine autoimmune oophoritis, epididymoorchitis, and gastritis induced by day 3 thymectomy. Immunopathology. Am. J. Pathol. 126, 293–302 (1987).
- Taguchi, O., Nishizuka, Y., Sakakura, T. & Kojima, A. Autoimmune oophoritis in thymectomized mice: detection of circulating antibodies against oocytes. Clin. Exp. Immunol. 40, 540–553 (1980).
- Nishizuka, Y. & Sakakura, T. Thymus and reproduction: sex-linked dysgenesia of the gonad after neonatal thymectomy in mice. Science 166, 753–755 (1969).
- Sakaguchi, S., Takahashi, T. & Nishizuka, Y. Study on cellular events in post-thymectomy autoimmune oophoritis in mice. II. Requirement of Lyt-1 cells in normal female mice for the prevention of oophoritis. J. Exp. Med. 156, 1577–1586 (1982).
- Sakaguchi, S., Sakaguchi, N., Asano, M., Itoh, M. & Toda, M. Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J. Immunol. Baltim. Md 1950 155, 1151–1164 (1995).
- Brunkow, M. E. et al. Disruption of a new forkhead/winged-helix protein, scurfin, results in the fatal lymphoproliferative disorder of the scurfy mouse. Nat. Genet. 27, 68–73 (2001).
- Bennett, C. L. et al. The immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) is caused by mutations of FOXP3. Nat. Genet. 27, 20–21 (2001).
- Wildin, R. S. et al. X-linked neonatal diabetes mellitus, enteropathy and endocrinopathy syndrome is the human equivalent of mouse scurfy. Nat. Genet. 27, 18–20 (2001).
- Hori, S., Nomura, T. & Sakaguchi, S. Control of regulatory T cell development by the transcription factor Foxp3. Science 299, 1057–1061 (2003).
- Khattri, R., Cox, T., Yasayko, S.-A. & Ramsdell, F. An essential role for Scurfin in CD4+CD25+ T regulatory cells. Nat. Immunol. 4, 337–342 (2003).
- Vignali, D. A. A., Collison, L. W. & Workman, C. J. How regulatory T cells work. Nat. Rev. Immunol. 8, 523–532 (2008).
- Viglietta, V., Baecher-Allan, C., Weiner, H. L. & Hafler, D. A. Loss of functional suppression by CD4+CD25+ regulatory T cells in patients with multiple sclerosis. J. Exp. Med. 199, 971–979 (2004).
- Ehrenstein, M. R. et al. Compromised Function of Regulatory T Cells in Rheumatoid Arthritis and Reversal by Anti-TNFα Therapy. J. Exp. Med. 200, 277–285 (2004).
- Nussbaum, L., Chen, Y. L. & Ogg, G. S. Role of regulatory T cells in psoriasis pathogenesis and treatment. Br. J. Dermatol. 184, 14–24 (2021).
- Lindley, S. et al. Defective Suppressor Function in CD4+CD25+ T-Cells From Patients With Type 1 Diabetes. Diabetes 54, 92–99 (2005).
- Klatzmann, D. & Abbas, A. K. The promise of low-dose interleukin-2 therapy for autoimmune and inflammatory diseases. Nat. Rev. Immunol. 15, 283–294 (2015).
- Bender, C. et al. A phase 2 randomized trial with autologous polyclonal expanded regulatory T cells in children with new-onset type 1 diabetes. Sci. Transl. Med. 16, eadn2404 (2024).
- Plitas, G. & Rudensky, A. Y. Regulatory T Cells in Cancer. Annu. Rev. Cancer Biol. 4, 459–477 (2020).
- Walsh, P. T., Taylor, D. K. & Turka, L. A. Tregs and transplantation tolerance. J. Clin. Invest. 114, 1398–1403 (2004).
